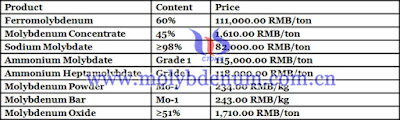
Molybdenum concentrate market in China has performed well recently. 40-45% molybdenum ore is concluded 60 tons at RMB 1,610/t; 45-50% molybdenum concentrate is concluded with price of RMB 1,635/t and quantity of 200 tons. Steel mills’ purchasing in large number stimulates ferromolybdenum manufacturers to enquiry. Under the background, molybdenum mines and traders raised their strong rised mentality.
Market participants’large demand for molybdenum concentrate guarantees the high price of the market. While the price of steel biddings fails to reach RMB 110,000/t, resulting ferromolybdenum manufacturers’cautious attitude towards purchasing on high-price molybdenum concentrate. In April, steel mills purchased large amount of ferro molybdenum, so ferro molybdenum manufacturers will mainly focus on production and thus the quotation would remain stable.
Prices of molybdenum products on Apr. 28, 2018
Picture of ferro molybdenum
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
2018年4月27日星期五
2018年4月26日星期四
Occurrence of Molybdenum
Molybdenum is the 54th most abundant element in the Earth's crust and the 25th most abundant element in the oceans, with an average of 10 parts per billion; it is the 42nd most abundant element in the Universe. The Russian Luna 24 mission discovered a molybdenum-bearing grain (1 × 0.6 µm) in a pyroxene fragment taken from Mare Crisium on the Moon.
The comparative rarity of molybdenum in the Earth's crust is offset by its concentration in a number of water-insoluble ores, often combined with sulfur, in the same way as copper, with which it is often found. Though molybdenum is found in such minerals as wulfenite (PbMoO4) and powellite (CaMoO4), the main commercial source of molybdenum is molybdenite (MoS2). Molybdenum is mined as a principal ore, and is also recovered as a byproduct of copper and tungsten mining.
Historically, the Knaben mine in southern Norway, opened in 1885, was the first dedicated molybdenum mine. It closed from 1973–2007, but is now reopened. Large mines in Colorado (such as the Henderson mine and the Climax mine) and in British Columbia yield molybdenite as their primary product, while many porphyry copper deposits such as the Bingham Canyon Mine in Utah and the Chuquicamata mine in northern Chile produce molybdenum as a byproduct of copper mining.
The world's production of molybdenum was 250,000 tonnes in 2011, the largest producers being China (94,000 t), United States (64,000 t), Chile (38,000 t), Peru (18,000 t) and Mexico (12,000 t). The total reserves are estimated at 10 million tonnes, and are mostly concentrated in China (4.3 mt), US (2.7 mt) and Chile (1.2 mt). By continent, 93% of world molybdenum production is about evenly split between North America, South America (mainly in Chile), and China. Europe and the rest of Asia (mostly Armenia, Russia, Iran and Mongolia) produce the remainder.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
The comparative rarity of molybdenum in the Earth's crust is offset by its concentration in a number of water-insoluble ores, often combined with sulfur, in the same way as copper, with which it is often found. Though molybdenum is found in such minerals as wulfenite (PbMoO4) and powellite (CaMoO4), the main commercial source of molybdenum is molybdenite (MoS2). Molybdenum is mined as a principal ore, and is also recovered as a byproduct of copper and tungsten mining.
Historically, the Knaben mine in southern Norway, opened in 1885, was the first dedicated molybdenum mine. It closed from 1973–2007, but is now reopened. Large mines in Colorado (such as the Henderson mine and the Climax mine) and in British Columbia yield molybdenite as their primary product, while many porphyry copper deposits such as the Bingham Canyon Mine in Utah and the Chuquicamata mine in northern Chile produce molybdenum as a byproduct of copper mining.
The world's production of molybdenum was 250,000 tonnes in 2011, the largest producers being China (94,000 t), United States (64,000 t), Chile (38,000 t), Peru (18,000 t) and Mexico (12,000 t). The total reserves are estimated at 10 million tonnes, and are mostly concentrated in China (4.3 mt), US (2.7 mt) and Chile (1.2 mt). By continent, 93% of world molybdenum production is about evenly split between North America, South America (mainly in Chile), and China. Europe and the rest of Asia (mostly Armenia, Russia, Iran and Mongolia) produce the remainder.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
2018年4月25日星期三
History of Molybdenum
Molybdenite—the principal ore from which molybdenum is now extracted—was previously known as molybdena. Molybdena was confused with and often utilized as though it were graphite. Like graphite, molybdenite can be used to blacken a surface or as a solid lubricant. Even when molybdena was distinguishable from graphite, it was still confused with the common lead ore PbS (now called galena); the name comes from Ancient Greek Μόλυβδος molybdos, meaning lead. (The Greek word itself has been proposed as a loanword from Anatolian Luvian and Lydian languages).
Although apparent deliberate alloying of molybdenum with steel in one 14th-century Japanese sword (mfd. ca. 1330) has been reported, that art was never employed widely and was later lost. In in the West in 1754, Bengt Andersson Qvist examined molybdenite and determined that it did not contain lead, and thus was not the same as galena.
By 1778 Swedish chemist Carl Wilhelm Scheele stated firmly that molybdena was (indeed) not galena nor graphite. Instead, Scheele went further and correctly proposed that molybdena was an ore of a distinct new element, named molybdenum for the mineral in which it resided, and from which it might be isolated. Peter Jacob Hjelm successfully isolated molybdenum by using carbon and linseed oil in 1781.
For about a century after its isolation, molybdenum had no industrial use, owing to its relative scarcity, difficulty extracting the pure metal, and the immaturity of appropriate metallurgical techniques. Early molybdenum steel alloys showed great promise in their increased hardness, but efforts to manufacture them on a large scale were hampered by inconsistent results and a tendency toward brittleness and recrystallization.
In 1906, William D. Coolidge filed a patent for rendering molybdenum ductile, leading to its use as a heating element for high-temperature furnaces and as a support for tungsten-filament light bulbs; oxide formation and degradation require that molybdenum be physically sealed or held in an inert gas. In 1913, Frank E. Elmore developed a flotation process to recover molybdenite from ores; flotation remains the primary isolation process.
During the first World War, demand for molybdenum spiked; it was used both in armor plating and as a substitute for tungsten in high speed steels. Some British tanks were protected by 75 mm (3 in) manganese steel plating, but this proved to be ineffective. The manganese steel plates were replaced with 25 mm (1 in) molybdenum-steel plating allowing for higher speed, greater maneuverability, and better protection.
The Germans also used molybdenum-doped steel for heavy artillery. This was because traditional steel melted at the heat produced by enough gunpowder to launch a one ton shell. After the war, demand plummeted until metallurgical advances allowed extensive development of peacetime applications. In World War II, molybdenum again saw strategic importance as a substitute for tungsten in steel alloys.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
Although apparent deliberate alloying of molybdenum with steel in one 14th-century Japanese sword (mfd. ca. 1330) has been reported, that art was never employed widely and was later lost. In in the West in 1754, Bengt Andersson Qvist examined molybdenite and determined that it did not contain lead, and thus was not the same as galena.
By 1778 Swedish chemist Carl Wilhelm Scheele stated firmly that molybdena was (indeed) not galena nor graphite. Instead, Scheele went further and correctly proposed that molybdena was an ore of a distinct new element, named molybdenum for the mineral in which it resided, and from which it might be isolated. Peter Jacob Hjelm successfully isolated molybdenum by using carbon and linseed oil in 1781.
For about a century after its isolation, molybdenum had no industrial use, owing to its relative scarcity, difficulty extracting the pure metal, and the immaturity of appropriate metallurgical techniques. Early molybdenum steel alloys showed great promise in their increased hardness, but efforts to manufacture them on a large scale were hampered by inconsistent results and a tendency toward brittleness and recrystallization.
In 1906, William D. Coolidge filed a patent for rendering molybdenum ductile, leading to its use as a heating element for high-temperature furnaces and as a support for tungsten-filament light bulbs; oxide formation and degradation require that molybdenum be physically sealed or held in an inert gas. In 1913, Frank E. Elmore developed a flotation process to recover molybdenite from ores; flotation remains the primary isolation process.
During the first World War, demand for molybdenum spiked; it was used both in armor plating and as a substitute for tungsten in high speed steels. Some British tanks were protected by 75 mm (3 in) manganese steel plating, but this proved to be ineffective. The manganese steel plates were replaced with 25 mm (1 in) molybdenum-steel plating allowing for higher speed, greater maneuverability, and better protection.
The Germans also used molybdenum-doped steel for heavy artillery. This was because traditional steel melted at the heat produced by enough gunpowder to launch a one ton shell. After the war, demand plummeted until metallurgical advances allowed extensive development of peacetime applications. In World War II, molybdenum again saw strategic importance as a substitute for tungsten in steel alloys.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
2018年4月24日星期二
Molybdenum Compounds
Molybdenum is a transition metal with an electronegativity of 2.16 on the Pauling scale and a standard atomic weight of 95.96 g/mol. It does not visibly react with oxygen or water at room temperature, and the bulk oxidation occurs at temperatures above 600 °C, resulting in molybdenum trioxide: 2 Mo + 3 O2 → 2 MoO3
The trioxide is volatile and sublimates at high temperatures. This prevents formation of a continuous protective oxide layer, which would stop the bulk oxidation of metal. Molybdenum has several oxidation states, the most stable being +4 and +6 (bolded in the table). The chemistry and the compounds show more similarity to those of tungsten than that of chromium. An example is the instability of molybdenum(III) and tungsten(III) compounds as compared with the stability of the chromium(III) compounds. The highest oxidation state is common in the molybdenum(VI) oxide (MoO3), whereas the normal sulfur compound is molybdenum disulfide MoS2.
Molybdenum(VI) oxide is soluble in strong alkaline water, forming molybdates (MoO42−). Molybdates are weaker oxidants than chromates, but they show a similar tendency to form complex oxyanions by condensation at lower pH values, such as [Mo7O24]6− and [Mo8O26]4−. Polymolybdates can incorporate other ions into their structure, forming polyoxometalates. The dark-blue phosphorus-containing heteropolymolybdate P[Mo12O40]3− is used for the spectroscopic detection of phosphorus. The broad range of oxidation states of molybdenum is reflected in various molybdenum chlorides:
Molybdenum(II) chloride MoCl2 (yellow solid)
Molybdenum(IV) chloride MoCl4 (black solid)Molybdenum(III) chloride MoCl3 (dark red solid)
Molybdenum(V) chloride MoCl5 (dark green solid)
Molybdenum(VI) chloride MoCl6 (brown solid)
The structure of the MoCl2 is composed of Mo6Cl84+ clusters with four chloride ions to compensate the charge. Like chromium and some other transition metals, molybdenum is able to form quadruple bonds, such as in Mo2(CH3COO)4. This compound can be transformed into Mo2Cl84−, which also has a quadruple bond. The oxidation state 0 is possible with carbon monoxide as ligand, such as in molybdenum hexacarbonyl, Mo(CO)6.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
The trioxide is volatile and sublimates at high temperatures. This prevents formation of a continuous protective oxide layer, which would stop the bulk oxidation of metal. Molybdenum has several oxidation states, the most stable being +4 and +6 (bolded in the table). The chemistry and the compounds show more similarity to those of tungsten than that of chromium. An example is the instability of molybdenum(III) and tungsten(III) compounds as compared with the stability of the chromium(III) compounds. The highest oxidation state is common in the molybdenum(VI) oxide (MoO3), whereas the normal sulfur compound is molybdenum disulfide MoS2.
Molybdenum(VI) oxide is soluble in strong alkaline water, forming molybdates (MoO42−). Molybdates are weaker oxidants than chromates, but they show a similar tendency to form complex oxyanions by condensation at lower pH values, such as [Mo7O24]6− and [Mo8O26]4−. Polymolybdates can incorporate other ions into their structure, forming polyoxometalates. The dark-blue phosphorus-containing heteropolymolybdate P[Mo12O40]3− is used for the spectroscopic detection of phosphorus. The broad range of oxidation states of molybdenum is reflected in various molybdenum chlorides:
Molybdenum(II) chloride MoCl2 (yellow solid)
Molybdenum(IV) chloride MoCl4 (black solid)Molybdenum(III) chloride MoCl3 (dark red solid)
Molybdenum(V) chloride MoCl5 (dark green solid)
Molybdenum(VI) chloride MoCl6 (brown solid)
The structure of the MoCl2 is composed of Mo6Cl84+ clusters with four chloride ions to compensate the charge. Like chromium and some other transition metals, molybdenum is able to form quadruple bonds, such as in Mo2(CH3COO)4. This compound can be transformed into Mo2Cl84−, which also has a quadruple bond. The oxidation state 0 is possible with carbon monoxide as ligand, such as in molybdenum hexacarbonyl, Mo(CO)6.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
2018年4月23日星期一
Isotopes of Molybdenum
There are 35 known isotopes of molybdenum, ranging in atomic mass from 83 to 117, as well as four metastable nuclear isomers. Seven isotopes occur naturally, with atomic masses of 92, 94, 95, 96, 97, 98, and 100. Of these naturally occurring isotopes, only molybdenum-100 is unstable.
Molybdenum-98 is the most abundant isotope, comprising 24.14% of all molybdenum. Molybdenum-100 has a half-life of about 1019 y and undergoes double beta decay into ruthenium-100. Molybdenum isotopes with mass numbers from 111 to 117 all have half-lives of approximately 150 ns. All unstable isotopes of molybdenum decay into isotopes of niobium, technetium, and ruthenium.
As also noted below, the most common isotopic molybdenum application involves molybdenum-99, which is a fission product. It is a parent radioisotope to the short-lived gamma-emitting daughter radioisotope technetium-99m, a nuclear isomer used in various imaging applications in medicine.[9] In 2008, the Delft University of Technology applied for a patent on the molybdenum-98-based production of molybdenum-99.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
Molybdenum-98 is the most abundant isotope, comprising 24.14% of all molybdenum. Molybdenum-100 has a half-life of about 1019 y and undergoes double beta decay into ruthenium-100. Molybdenum isotopes with mass numbers from 111 to 117 all have half-lives of approximately 150 ns. All unstable isotopes of molybdenum decay into isotopes of niobium, technetium, and ruthenium.
As also noted below, the most common isotopic molybdenum application involves molybdenum-99, which is a fission product. It is a parent radioisotope to the short-lived gamma-emitting daughter radioisotope technetium-99m, a nuclear isomer used in various imaging applications in medicine.[9] In 2008, the Delft University of Technology applied for a patent on the molybdenum-98-based production of molybdenum-99.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
2018年4月22日星期日
About Molybdenum
Molybdenum is a Group 6 chemical element with the symbol Mo and atomic number 42.
The name is from Neo-Latin Molybdaenum, from Ancient Greek Μόλυβδος molybdos, meaning lead, since its ores were confused with lead ores.[4] Molybdenum minerals have been known into prehistory, but the element was "discovered" (in the sense of differentiating it as a new entity from the mineral salts of other metals) in 1778 by Carl Wilhelm Scheele. The metal was first isolated in 1781 by Peter Jacob Hjelm.
Molybdenum does not occur naturally as a free metal on Earth, but rather in various oxidation states in minerals. The free element, which is a silvery metal with a gray cast, has the sixth-highest melting point of any element. It readily forms hard, stable carbides in alloys, and for this reason most of world production of the element (about 80%) is in making many types of steel alloys, including high strength alloys and superalloys.
Most molybdenum compounds have low solubility in water, but the molybdate ion MoO42− is soluble and forms when molybdenum-containing minerals are in contact with oxygen and water. Industrially, molybdenum compounds (about 14% of world production of the element) are used in high-pressure and high-temperature applications, as pigments and catalysts.
Molybdenum-containing enzymes are by far the most common catalysts used by some bacteria to break the chemical bond in atmospheric molecular nitrogen, allowing biological nitrogen fixation. At least 50 molybdenum-containing enzymes are now known in bacteria and animals, although only bacterial and cyanobacterial enzymes are involved in nitrogen fixation, and these nitrogenases contain molybdenum in a different form from the rest. Owing to the diverse functions of the various other types of molybdenum enzymes, molybdenum is a required element for life in all higher organisms (eukaryotes), though not in all bacteria.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
The name is from Neo-Latin Molybdaenum, from Ancient Greek Μόλυβδος molybdos, meaning lead, since its ores were confused with lead ores.[4] Molybdenum minerals have been known into prehistory, but the element was "discovered" (in the sense of differentiating it as a new entity from the mineral salts of other metals) in 1778 by Carl Wilhelm Scheele. The metal was first isolated in 1781 by Peter Jacob Hjelm.
Molybdenum does not occur naturally as a free metal on Earth, but rather in various oxidation states in minerals. The free element, which is a silvery metal with a gray cast, has the sixth-highest melting point of any element. It readily forms hard, stable carbides in alloys, and for this reason most of world production of the element (about 80%) is in making many types of steel alloys, including high strength alloys and superalloys.
Most molybdenum compounds have low solubility in water, but the molybdate ion MoO42− is soluble and forms when molybdenum-containing minerals are in contact with oxygen and water. Industrially, molybdenum compounds (about 14% of world production of the element) are used in high-pressure and high-temperature applications, as pigments and catalysts.
Molybdenum-containing enzymes are by far the most common catalysts used by some bacteria to break the chemical bond in atmospheric molecular nitrogen, allowing biological nitrogen fixation. At least 50 molybdenum-containing enzymes are now known in bacteria and animals, although only bacterial and cyanobacterial enzymes are involved in nitrogen fixation, and these nitrogenases contain molybdenum in a different form from the rest. Owing to the diverse functions of the various other types of molybdenum enzymes, molybdenum is a required element for life in all higher organisms (eukaryotes), though not in all bacteria.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
2018年4月11日星期三
TZM Alloy Slab Hot Rolled Cogging Process
At 1250 ~ 1350 ℃, proceeding hot rolled process on TZM alloy slab found under the process TZM alloy has lower yields, mainly due to the hot-rolled stratified, splitting and other defects. From this we will know that hot rolling process has a greater impact on the quality of TZM alloy slab. The experiment found that using low-temperature hot-rolled technology to produce TZM alloy has good performance and high yield.
Low temperature hot rolled temperature at 850 ~ 1000 ℃, the second pass deformation higher than 25%, the blank denser than 9.6g/cm can produce the best performance TZM alloy.
Low-temperature hot-rolled cogging process has three advantages:
1. Obtaining a fine grain structure so it can improve the dispersion of the second phase of impurities, changing the composition and morphology of the second phase, to improve process performance;
2. Promoting the formation of sub-structure, improving the dislocation density can achieve deformation strengthening, improving the strength of the alloy;
3. Deeping deformation and increasing the uniformity of deformation can reduce delamination and cracks.
The following main analyze the splittings influence of second pass deformation on TZM hot-rolled slab. When the second pass deformation is higher than 25%, the deformation of the slab is deeper, and it can reduce the effect of the middle metal tensioning stress. When the slab after first rolling the surface of the metal layer has already happen plastic deformation, and the middle metal didn’t happen plastic deformation, so it is still in the state of powder sintering. The middle metal surface suffers metal tensile stress.
Therefore, the amount of deformation of the second pass should higher than 25%, so that deformations will deeper. If the amount of deformation of the second pass is not large, the deformation is not deeper, than the middle metal will suffer two times tensile stress effect. When the tensile stress is large enough to destroy the inter-crystal bonding force, so the intergranular fracture will occurs, leading to macroscopic wall crack.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
Low temperature hot rolled temperature at 850 ~ 1000 ℃, the second pass deformation higher than 25%, the blank denser than 9.6g/cm can produce the best performance TZM alloy.
Low-temperature hot-rolled cogging process has three advantages:
1. Obtaining a fine grain structure so it can improve the dispersion of the second phase of impurities, changing the composition and morphology of the second phase, to improve process performance;
2. Promoting the formation of sub-structure, improving the dislocation density can achieve deformation strengthening, improving the strength of the alloy;
3. Deeping deformation and increasing the uniformity of deformation can reduce delamination and cracks.
The following main analyze the splittings influence of second pass deformation on TZM hot-rolled slab. When the second pass deformation is higher than 25%, the deformation of the slab is deeper, and it can reduce the effect of the middle metal tensioning stress. When the slab after first rolling the surface of the metal layer has already happen plastic deformation, and the middle metal didn’t happen plastic deformation, so it is still in the state of powder sintering. The middle metal surface suffers metal tensile stress.
Therefore, the amount of deformation of the second pass should higher than 25%, so that deformations will deeper. If the amount of deformation of the second pass is not large, the deformation is not deeper, than the middle metal will suffer two times tensile stress effect. When the tensile stress is large enough to destroy the inter-crystal bonding force, so the intergranular fracture will occurs, leading to macroscopic wall crack.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
2018年4月10日星期二
TZM Alloy’s Breaking Tenacity Researching
Due to TZM alloy has great high temperature performance so it play an important role in national economy and sciences technologies. TZM piercing point is an important tool for producing steel tube. It can drill stainless steel, heat resistant steel and rock drill steel and the server life is 300 times than steel mould. During drill process, the piercing point easy broke this is because at the heat and cool condition the piercing point suffer pressing, pulling, twisting and other complex stress which easy occurs cold hot fatigue crack with drill times increase the cracks extent lead to the piercing point broke.
Breaking tenacity is a aggregative indicator for alloy's strengthening and plasticity, researching breaking tenacity for alloy technics investigation improving, machine design, inspection and maintenance is essential part. So researching breaking tenacity for explore new production technology and heating-treating process has significant meaning. The research data are as following.
1. TZM alloy after 1200℃vacuum treatment the breaking tenacity KIC valve is higher than sintering's.
2. After 1300℃, 1400 ℃ vacuum treatment and 1300℃, 1400℃,1500℃ hydrogen treatment the breaking tenacity KIC valve is lower than sintering's.
3. TZM alloy breaking mainly because the component has micro-crack, cavity and impurity so the raw material powder's properties and purity control should very strict.
4. TZM alloy at 1300℃ employ heat-treatment can't improve alloy's breaking tenacity. Only at 1200℃ vacuum treatment can improve breaking tenacity and the properties of alloy improves as well.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
Breaking tenacity is a aggregative indicator for alloy's strengthening and plasticity, researching breaking tenacity for alloy technics investigation improving, machine design, inspection and maintenance is essential part. So researching breaking tenacity for explore new production technology and heating-treating process has significant meaning. The research data are as following.
1. TZM alloy after 1200℃vacuum treatment the breaking tenacity KIC valve is higher than sintering's.
2. After 1300℃, 1400 ℃ vacuum treatment and 1300℃, 1400℃,1500℃ hydrogen treatment the breaking tenacity KIC valve is lower than sintering's.
3. TZM alloy breaking mainly because the component has micro-crack, cavity and impurity so the raw material powder's properties and purity control should very strict.
4. TZM alloy at 1300℃ employ heat-treatment can't improve alloy's breaking tenacity. Only at 1200℃ vacuum treatment can improve breaking tenacity and the properties of alloy improves as well.
If you have any inquiry of molybdenum products, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
2018年4月8日星期日
Hot-dip Aluminized Coating on TZM Alloy
TZM alloy(Mo-0.5 Ti-0.08 Zr-0.02(wt%))is kind of a high temperature alloy of molybdenum base alloy which are widely used in high temperature nuclear reactor, aviation, electricity generation and as the high temperature resistance material and structural material of chemical industry. However, alloy in the high temperature is easy oxidation and so it will limit alloy's application field. After studies found when the temperature higher than 540℃ the molybdenum of TZM alloy will oxidation quickly to form non-protection MoO3 oxide layer. This oxide layer will volatilize at 750℃ and make molybdenum matrix oxide and volatilize which is bad for alloy’s properties.
Using hot-dip aluminized coating technique to make aluminized layer on TZM alloy surface to improve high temperature oxidation resistance. The hot-dip temperature is 730℃and hot-dip time is 3min or 5min. Clad layer is combine with surface aluminized layer and inner alloy layer and the inner alloy layer is mainly Al4Mo and Al5Mo phase. Besides, the micro-hardness of inner alloy layer reach at HV760. The clad layer and substrate is metallurgical bonding. What’s more, aluminized layer has good high temperature oxidation resistance property.
If you have any inquiry of ZTM alloy sheet, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
Using hot-dip aluminized coating technique to make aluminized layer on TZM alloy surface to improve high temperature oxidation resistance. The hot-dip temperature is 730℃and hot-dip time is 3min or 5min. Clad layer is combine with surface aluminized layer and inner alloy layer and the inner alloy layer is mainly Al4Mo and Al5Mo phase. Besides, the micro-hardness of inner alloy layer reach at HV760. The clad layer and substrate is metallurgical bonding. What’s more, aluminized layer has good high temperature oxidation resistance property.
If you have any inquiry of ZTM alloy sheet, please feel free to contact us:
Email: sales@chinatungsten.com/sales@xiamentungsten.com
Tel.: +86 592 5129696/+86 592 5129595
Fax: +86 592 5129797
Scan QR code of Chinatungsten Wechat platform to follow up the daily latest price and market of tungsten, molybdenum and rare earth.
订阅:
评论 (Atom)